|
(a) This section applies to scented or scented deodorized menstrual tampons as identified in § 884.5460 and unscented menstrual tampons as identified in § 884.5470 of this chapter.
(b) Data show that toxic shock syndrome (TSS), a rare but serious and sometimes fatal disease, is associated with the use of menstrual tampons. To protect the public and to minimize the serious adverse effects of TSS, menstrual tampons shall be labeled as set forth in paragraphs (c), (d), and (e) of this section and tested for absorbency as set forth in paragraph (f) of this section.
(c) If the information specified in paragraph (d) of this section is to be included as a package insert, the following alert statement shall appear prominently and legibly on the package label:
Attention: Tampons are associated with Toxic Shock Syndrome (TSS). TSS is a rare but serious disease that may cause death. Read and save the enclosed information.
(d) The labeling of menstrual tampons shall contain the following consumer information prominently and legibly, in such terms as to render the information likely to be read and understood by the ordinary individual under customary conditions of purchase and use:
(1)(i) Warning signs of TSS, e.g., sudden fever (usually 102deg. or more) and vomiting, diarrhea, fainting or near fainting when standing up, dizziness, or a rash that looks like a sunburn;
(ii) What to do if these or other signs of TSS appear, including the need to remove the tampon at once and seek medical attention immediately;
(2) The risk of TSS to all women using tampons during their menstrual period, especially the reported higher risks to women under 30 years of age and teenage girls, the estimated incidence of TSS of 1 to 17 per 100,000 menstruating women and girls per year, and the risk of death from contracting TSS;
(3) The advisability of using tampons with the minimum absorbency needed to control menstrual flow in order to reduce the risk of contracting TSS;
(4) Avoiding the risk of getting tampon-associated TSS by not using tampons, and reducing the risk of getting TSS by alternating tampon use with sanitary napkin use during menstrual periods; and
(5) The need to seek medical attention before again using tampons if TSS warning signs have occurred in the past, or if women have any questions about TSS or tampon use.
(e) The statements required by paragraph (e) of this section shall be prominently and legibly placed on the package label of menstrual tampons in conformance with section 502(c) of the Federal Food, Drug, and Cosmetic Act (the act) (unless the menstrual tampons are exempt under paragraph (g) of this section).
(1) Menstrual tampon package labels shall bear one of the following absorbency terms representing the absorbency of the production run, lot, or batch as measured by the test described in paragraph (f)(2) of this section;
| Ranges of absorbency in grams
1
| Corresponding term of absorbency
|
|---|
| 6 and under | Light absorbency
| | 6 to 9 | Regular absorbency
| | 9 to 12 | Super absorbency
| | 12 to 15 | Super plus absorbency
| | 15 to 18 | Ultra absorbency
| | Above 18 | No term
|
(2) The package label shall include an explanation of the ranges of absorbency and a description of how consumers can use a range of absorbency, and its corresponding absorbency term, to make comparisons of absorbency of tampons to allow selection of the tampons with the minimum absorbency needed to control menstrual flow in order to reduce the risk of contracting TSS.
(f) A manufacturer shall measure the absorbency of individual tampons using the test method specified in paragraph (f)(2) of this section and calculate the mean absorbency of a production run, lot, or batch by rounding to the nearest 0.1 gram.
(1) A manufacturer shall design and implement a sampling plan that includes collection of probability samples of adequate size to yield consistent tolerance intervals such that the probability is 90 percent that at least 90 percent of the absorbencies of individual tampons within a brand and type are within the range of absorbency stated on the package label.
(2) In the absorbency test, an unlubricated condom, with tensile strength between 17 Mega Pascals (MPa) and 30 MPa, as measured according to the procedure in the American Society for Testing and Materials (ASTM) D 3492-97, "Standard Specification for Rubber Contraceptives (Male Condoms)"
1
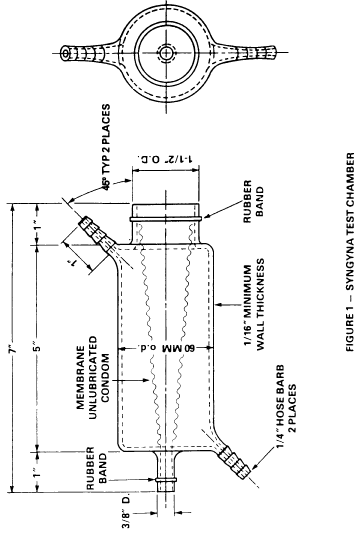
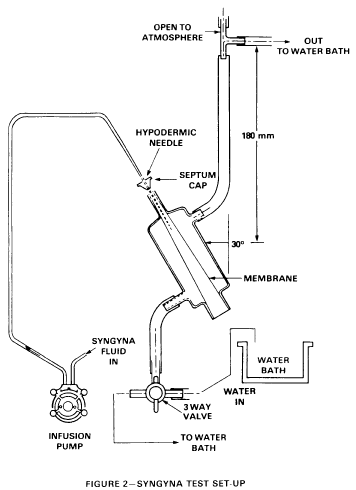
for determining tensile strength, which is incorporated by reference in accordance with 5 U.S.C. 552(a), is attached to the large end of a glass chamber (or a chamber made from hard transparent plastic) with a rubber band (see figure 1) and pushed through the small end of the chamber using a smooth, finished rod. The condom is pulled through until all slack is removed. The tip of the condom is cut off and the remaining end of the condom is stretched over the end of the tube and secured with a rubber band. A preweighed (to the nearest 0.01 gram) tampon is placed within the condom membrane so that the center of gravity of the tampon is at the center of the chamber. An infusion needle (14 gauge) is inserted through the septum created by the condom tip until it contacts the end of the tampon. The outer chamber is filled with water pumped from a temperature-controlled waterbath to maintain the average temperature at 27+/-1 deg.C. The water returns to the waterbath as shown in figure 2. Syngyna fluid (10 grams sodium chloride, 0.5 gram Certified Reagent Acid Fushsin, 1,000 milliliters distilled water) is then pumped through the infusion needle at a rate of 50 milliliters per hour. The test shall be terminated when the tampon is saturated and the first drop of fluid exits the apparatus. (The test result shall be discarded if fluid is detected in the folds of the condom before the tampon is saturated). The water is then drained and the tampon is removed and immediately weighed to the nearest 0.01 gram. The absorbency of the tampon is determined by subtracting its dry weight from this value. The condom shall be replaced after 10 tests or at the end of the day during which the condom is used in testing, whichever occurs first.
(3) The Food and Drug Administration may permit the use of an absorbency test method different from the test method specified in this section if each of the following conditions is met:
(i) The manufacturer presents evidence, in the form of a citizen petition submitted in accordance with the requirements of § 10.30 of this chapter, demonstrating that the alternative test method will yield results that are equivalent to the results yielded by the test method specified in this section; and
(ii) FDA approves the method and has published notice of its approval of the alternative test method in the Federal Register.
(g) Any menstrual tampon intended to be dispensed by a vending machine is exempt from the requirements of this section.
(h) Any menstrual tampon that is not labeled as required by paragraphs (c), (d), and (e) of this section and that is initially introduced or initially delivered for introduction into commerce after March 1, 1990, is misbranded under sections 201(n), 502 (a) and (f) of the act.
1The Director of the Federal Register approves this incorporation by reference in accordance with 5 U.S.C. 552(a) and 1 CFR part 51. You may obtain a copy from the American Society for Testing and Materials International, 100 Barr Harbor Dr., P.O. Box C700, West Conshohocken, PA 19428-2959, 610-832-9578, www.astm.org. You may inspect a copy at the FDA Main Library, 10903 New Hampshire Ave., Bldg. 2, 3d floor, Silver Spring, MD 20993-0002, 301-796-2039, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-2139, or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html. [47 FR 26989, June 22, 1982, as amended at 54 FR 43771, Oct. 26, 1989; 55 FR 17600, Apr. 26, 1990; 65 FR 3586, Jan. 24, 2000; 65 FR 44436, July 18, 2000; 65 FR 62284, Oct. 18, 2000; 69 FR 18803, Apr. 9, 2004; 69 FR 52171, Aug. 25, 2004; 75 FR 20914, Apr. 22, 2010]
|