|
As used in this subchapter J:
(a) Accidental radiation occurrence means a single accidental event or series of accidental events that has/have resulted in injurious or potentially injurious exposure of any person to electronic product radiation as a result of the manufacturing, testing, or use of an electronic product.
(b) Act means the Federal Food, Drug, and Cosmetic Act (21 U.S.C. 360hh-360ss).
(c) Chassis family means a group of one or more models with all of the following common characteristics:
(1) The same circuitry in the high voltage, horizontal oscillator, and power supply sections;
(2) The same worst component failures;
(3) The same type of high voltage hold-down or safety circuits; and
(4) The same design and installation.
(d) Commerce means:
(1) Commerce between any place in any State and any place outside thereof, and
(2) Commerce wholly within the District of Columbia.
(e) Component, for the purposes of this part, means an essential functional part of a subassembly or of an assembled electronic product, and which may affect the quantity, quality, direction, or radiation emission of the finished product.
(f) Dealer means a person engaged in the business of offering electronic products for sale to purchasers, without regard to whether such person is or has been primarily engaged in such business, and includes persons who offer such products for lease or as prizes or awards.
(g) Director means the Director of the Center for Devices and Radiological Health.
(h) Distributor means a person engaged in the business of offering electronic products for sale to dealers, without regard to whether such person is or has been primarily or customarily engaged in such business.
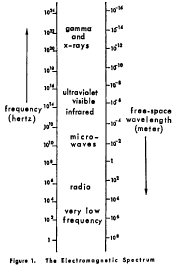
(i) Electromagnetic radiation includes the entire electromagnetic spectrum of radiation of any wavelength. The electromagnetic spectrum illustrated in figure 1 includes, but is not limited to, gamma rays, x-rays, ultra-violet, visible, infrared, microwave, radiowave, and low frequency radiation.
(j) Electronic product means:
(1) Any manufactured or assembled product which, when in operation:
(i) Contains or acts as part of an electronic circuit and
(ii) Emits (or in the absence of effective shielding or other controls would emit) electronic product radiation, or
(2) Any manufactured or assembled article that is intended for use as a component, part, or accessory of a product described in paragraph (j)(1) of this section and which, when in operation, emits (or in the absence of effective shielding or other controls would emit) such radiation.
(k) Electronic product radiation means:
(1) Any ionizing or nonionizing electromagnetic or particulate radiation, or
(2) Any sonic, infrasonic, or ultrasonic wave that is emitted from an electronic product as the result of the operation of an electronic circuit in such product.
(l) Federal standard means a performance standard issued pursuant to section 534 of the Federal Food, Drug, and Cosmetic Act.
(m) Infrasonic, sonic (or audible) and ultrasonic waves refer to energy transmitted as an alteration (pressure, particle displacement or density) in a property of an elastic medium (gas, liquid or solid) that can be detected by an instrument or listener.
(n) Manufacturer means any person engaged in the business of manufacturing, assembling, or importing electronic products.
(o) Model means any identifiable, unique electronic product design, and refers to products having the same structural and electrical design characteristics and to which the manufacturer has assigned a specific designation to differentiate between it and other products produced by that manufacturer.
(p) Model family means products having similar design and radiation characteristics but different manufacturer model numbers.
(q) Modified model means a product that is redesigned so that actual or potential radiation emission, the manner of compliance with a standard, or the manner of radiation safety testing is affected.
(r) Particulate radiation is defined as:
(1) Charged particles, such as protons, electrons, alpha particles, or heavy particles, which have sufficient kinetic energy to produce ionization or atomic or electron excitation by collision, electrical attractions or electrical repulsion; or
(2) Uncharged particles, such as neutrons, which can initiate a nuclear transformation or liberate charged particles having sufficient kinetic energy to produce ionization or atomic or electron excitation.
(s) Purchaser means the first person who, for value, or as an award or prize, acquires an electronic product for purposes other than resale, and includes a person who leases an electronic product for purposes other than subleasing.
(t) State means a State, the District of Columbia, the Commonwealth of Puerto Rico, the Virgin Islands, Guam, and American Samoa.
[60 FR 48380, Sept. 19, 1995; 61 FR 13422, Mar. 27, 1996, as amended at 88 FR 3652, Jan. 20, 2023]
|