|
|
| [Code of Federal Regulations] |
| [Title 21, Volume 3] |
| [CITE: 21CFR172] |
TITLE 21--FOOD AND DRUGS CHAPTER I--FOOD AND DRUG ADMINISTRATION DEPARTMENT OF HEALTH AND HUMAN SERVICES
SUBCHAPTER B - FOOD FOR HUMAN CONSUMPTION (CONTINUED)
| |
| PART 172 | FOOD ADDITIVES PERMITTED FOR DIRECT ADDITION TO FOOD FOR HUMAN CONSUMPTION |
|
|
|
|
Subpart B - Food Preservatives
|
|
Anoxomer as identified in this section may be safely used in accordance with the following conditions:
(a) Anoxomer is 1,4-benzenediol, 2-(1,1-dimethylethyl)-polymer with diethenylbenzene, 4-(1,1-dimethyl-ethyl)phenol, 4- methoxyphenol, 4,4'-(1-methylethylidene)bis(phenol) and 4-methylphenol (CAS Reg. No. 60837-57-2) prepared by condensation polymerization of divinylbenzene (m - and p -) with tert -butylhydroquinone, tert -butylphenol, hydroxyanisole, p -cresol and 4,4'-isopropylidenediphenol.
(b) The polymeric antioxidant meets the following specifications:
(1) Polymer, not less than 98.0 percent as determined by an ultraviolet method entitled "Ultraviolet Assay, "1982, which is incorporated by reference. Copies are available from the Office of Food Additive Safety (HFS-200), Center for Food Safety and Applied Nutrition, Food and Drug Administration, 5001 Campus Dr., College Park, MD 20740, 240-402-1200, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030, or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html.
(2) Molecular weight: Total monomers, dimers and trimers below 500 not more than 1 percent as determined by a method entitled "Low Molecular Weight Anoxomer Analysis," 1982, which is incorporated by reference. Copies are available from the Office of Food Additive Safety (HFS-200), Center for Food Safety and Applied Nutrition, Food and Drug Administration, 5001 Campus Dr., College Park, MD 20740, 240-402-1200, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030, or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html.
(3) Phenol content: Not less than 3.2 milliequivalent/gram and not more than 3.8 milliequivalent/gram as determined by a method entitled "Total Phenols," 1982, which is incorporated by reference. Copies are available from the Office of Food Additive Safety (HFS-200), Center for Food Safety and Applied Nutrition, Food and Drug Administration, 5001 Campus Dr., College Park, MD 20740, 240-402-1200, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030, or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html.
(4) Heavy metals as lead (as Pb), not more than 10 parts per million. Arsenic (as As), not more than 3 parts per million. Mercury (as Hg), not more than 1 part per million.
(c) Anoxomer may be safely used as an antioxidant in food at a level of not more than 5,000 parts per million based on fat and oil content of the food.
[48 FR 18798, Apr. 26, 1983, as amended at 54 FR 24896, June 12, 1989; 88 FR 17719, Mar. 24, 2023]
|
|
|
The food additive BHA (butylated hydroxyanisole) alone or in combination with other antioxidants permitted in food for human consumption in this subpart B may be safely used in or on specified foods, as follows:
(a) The BHA meets the following specification:
Assay (total BHA), 98.5 percent minimum. Melting point 48 deg.C minimum.
(b) The BHA is used alone or in combination with BHT, as an antioxidant in foods, as follows:
| Food
| Limitations (total BHA and BHT) parts per million
|
|---|
| Dehydrated potato shreds | 50
| | Active dry yeast |
1 1,000
| | Beverages and desserts prepared from dry mixes |
1 2
| | Dry breakfast cereals | 50
| | Dry diced glazed fruit |
1 32
| | Dry mixes for beverages and desserts |
1 90
| | Emulsion stabilizers for shortenings | 200
| | Potato flakes | 50
| | Potato granules | 10
| | Sweet potato flakes | 50
|
(c) To assure safe use of the additive:
(1) The label of any market package of the additive shall bear, in addition to the other information required by the Act, the name of the additive.
(2) When the additive is marketed in a suitable carrier, in addition to meeting the requirement of paragraph (c)(1) of this section, the label shall declare the percentage of the additive in the mixture.
(3) The label or labeling of dry mixes for beverages and desserts shall bear adequate directions for use to provide that beverages and desserts prepared from the dry mixes contain no more than 2 parts per million BHA.
|
|
|
The food additive BHT (butylated hydroxytoluene), alone or in combination with other antioxidants permitted in this subpart B may be safely used in or on specified foods, as follows:
(a) The BHT meets the following specification: Assay (total BHT) 99 percent minimum.
(b) The BHT is used alone or in combination with BHA, as an antioxidant in foods, as follows:
| Food
| Limitations (total BHA and BHT) parts per million
|
|---|
| Dehydrated potato shreds | 50
| | Dry breakfast cereals | 50
| | Emulsion stabilizers for shortenings | 200
| | Potato flakes | 50
| | Potato granules | 10
| | Sweetpotato flakes | 50 |
(c) To assure safe use of the additive:
(1) The label of any market package of the additive shall bear, in addition to the other information required by the Act, the name of the additive.
(2) When the additive is marketed in a suitable carrier, in addition to meeting the requirement of paragraph (c)(1) of this section, the label shall declare the percentage of the additive in the mixture.
|
|
|
Sec. 172.120 Calcium disodium EDTA.
|
|
The food additive calcium disodium EDTA (calcium disodium ethylene-diaminetetraacetate) may be safely used in designated foods for the purposes and in accordance with the conditions prescribed, as follows:
(a) The additive contains a minimum of 99 percent by weight of either the dihydrate C10H12O8N2CaNa2-2H2O or the trihydrate C10H12O8N2CaNa2-3H2O, or any mixture of the two.
(b) It is used or intended for use as follows:
(1) Alone, in the following foods at not to exceed the levels prescribed, calculated as the anhydrous compound:
| Food
| Limitation (parts per million)
| Use
|
|---|
| Cabbage, pickled | 220 | Promote color, flavor, and texture retention.
| | Canned carbonated soft drinks | 33 | Promote flavor retention.
| | Canned white potatoes | 110 | Promote color retention.
| | Clams (cooked canned) | 340 | Promote color retention.
| | Crabmeat (cooked canned) | 275 | Retard struvite formation; promote color retention.
| | Cucumbers pickled | 220 | Promote color, flavor, and texture retention.
| | Distilled alcoholic beverages | 25 | Promote stability of color, flavor, and/or product clarity.
| | Dressings, nonstandardized | 75 | Preservative.
| | Dried lima beans (cooked canned) | 310 | Promote color retention.
| | Egg product that is hard-cooked and consists, in a cylindrical shape, of egg white with an inner core of egg yolk |
1 200 | Preservative.
| | Fermented malt beverages | 25 | Antigushing agent.
| | French dressing | 75 | Preservative.
| | Legumes (all cooked canned, other than dried lima beans, pink beans, and red beans) | 365 | Promote color retention.
| | Mayonnaise | 75 | Do.
| | Mushrooms (cooked canned) | 200 | Promote color retention.
| | Oleomargarine | 75 | Preservative.
| | Pecan pie filling | 100 | Promote color retention.
| | Pink beans (cooked canned) | 165 | Promote color retention.
| | Potato salad | 100 | Preservative.
| | Processed dry pinto beans | 800 | Promote color retention.
| | Red beans (cooked canned) | 165 | Promote color retention.
| | Salad dressing | 75 | Preservative.
| | Sandwich spread | 100 | Do.
| | Sauces | 75 | Do.
| | Shrimp (cooked canned) | 250 | Retard struvite formation; promote color retention.
| | Spice extractives in soluble carriers | 60 | Promote color and flavor retention.
| | Spreads, artificially colored and lemon-flavored or orange-flavored | 100 | Promote color retention.
|
(2) With disodium EDTA (disodium ethylenediaminetetraacetate) in the following foods at not to exceed, in combination, the levels prescribed, calculated as anhydrous C10H12O8N2CaNa2:
| Food
| Limitation (parts per million)
| Use
|
|---|
| Dressings, nonstandardized | 75 | Preservative.
| | French dressing | 75 | Do.
| | Mayonnaise | 75 | Do.
| | Salad dressing | 75 | Do.
| | Sandwich spread | 100 | Do.
| | Sauces | 75 | Do. |
(c) To assure safe use of the additive:
(1) The label and labeling of the additive container shall bear, in addition to the other information required by the Act, the name of the additive.
(2) The label or labeling of the additive container shall bear adequate use directions to provide a final food product that complies with the limitations provided in paragraph (b) of this section.
(d) In the standardized foods listed in paragraph (b) of this section, the additives are used only in compliance with the applicable standards of identity for such foods.
[42 FR 14491, Mar. 15, 1977, as amended at 48 FR 10815, Mar. 15, 1983; 58 FR 52222, Oct. 7, 1993; 60 FR 33710, June 29, 1995; 65 FR 48379, Aug. 8, 2000]
|
|
|
Sec. 172.130 Dehydroacetic acid.
|
|
The food additive dehydroacetic acid and/or its sodium salt may be safely used in accordance with the following prescribed conditions:
(a) The food additive meets the following specifications:
Dehydroacetic acid: Melting point, 109 deg.C-111 deg.C; assay, minimum 98 percent (dry basis).
Sodium salt of dehydroacetic acid: Assay, minimum 98 percent (dry basis).
(b) It is used or intended for use as a preservative for cut or peeled squash, and is so used that no more than 65 parts per million expressed as dehydroacetic acid remains in or on the prepared squash.
(c) The label or labeling of any package of the additive intended for use in food shall bear adequate directions for use to insure compliance with this section.
|
|
|
Sec. 172.133 Dimethyl dicarbonate.
|
|
Dimethyl dicarbonate (CAS Reg. No. 4525-33-1) may be safely used in food in accordance with the following prescribed conditions:
(a) The additive meets the following specifications:
(1) The additive has a purity of not less than 99.8 percent as determined by the following titration method:
principles of method
Dimethyl dicarbonate (DMDC) is mixed with excess diisobutylamine with which it reacts quantitatively. The excess amine is backtitrated with acid.
apparatus
250-milliliter (mL) Beaker
100-mL Graduate cylinder
25-mL Pipette
10-mL Burette (automatic, e.g., Metrohm burette)
Stirrer
Device for potentiometric titration
Reference electrode
Glass electrode
reagents
Acetone, analytical-grade
Solution of 1 N diisobutylamine in chlorobenzene, distilled
1 N Acetic Acid
procedure
Accurately weigh in about 2 grams of the sample (W) and dissolve in 100 mL acetone. Add accurately 25 mL of the 1 N diisobutylamine solution by pipette and allow to stand for 5 minutes. Subsequently, titrate the reaction mixture potentiometrically with 1 N hydrochloric acid (consumption=a mL) while stirring. For determining the blank consumption, carry out the analysis without a sample (consumption=b mL).
calculation
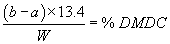
(2) The additive contains not more than 2,000 ppm (0.2 percent) dimethyl carbonate as determined by a method entitled "Gas Chromatography Method for Dimethyl Carbonate Impurity in Dimethyl Dicarbonate," which is incorporated by reference in accordance with 5 U.S.C. 552(a). Copies are available from the Office of Food Additive Safety (HFS-200), Center for Food Safety and Applied Nutrition, Food and Drug Administration, 5001 Campus Dr., College Park, MD 20740, 240-402-1200, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030, or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html.
(b) The additive is used or intended for use as a microbial control agent in the following beverages under normal circumstances of bottling, canning, or other forms of final packaging, where the viable microbial load has been reduced to 500 microorganisms per milliliter or less by current good manufacturing practices such as heat treatment, filtration, or other technologies prior to the use of dimethyl dicarbonate:
(1) In wine, dealcoholized wine, and low alcohol wine in an amount not to exceed 200 parts per million.
(2) In ready-to-drink teas in an amount not to exceed 250 parts per million.
(3) In carbonated or noncarbonated, nonjuice-containing (less than or equal to 1 percent juice), flavored or unflavored beverages containing added electrolytes (5-20 milliequivalents/liter sodium ion (Na + ) and 3-7 milliequivalents/liter potassium ion (K + )) in an amount not to exceed 250 parts per million.
(4) In carbonated, dilute beverages containing juice, fruit flavor, or both, with juice content not to exceed 50 percent, in an amount not to exceed 250 parts per million.
(c) To ensure the safe use of the food additive, the label of the package containing the additive shall bear, in addition to other information required by the Federal Food, Drug, and Cosmetic Act:
(1) The name of the additive "dimethyl dicarbonate."
(2) The intended use of the additive.
(3) Adequate directions for use to ensure compliance with this section.
For adding the diisobutylamine solution, always use the same pipette and wait for a further three drops to fall when the flow has stopped. Note: [53 FR 41329, Oct. 21, 1988, as amended at 58 FR 6091, Jan. 26, 1993; 59 FR 5319, Feb. 4, 1994; 61 FR 14245, Apr. 1, 1996; 61 FR 26788, May 29, 1996; 66 FR 13653, Mar. 7, 2001; 88 FR 17719, Mar. 24, 2023]
|
|
|
Sec. 172.135 Disodium EDTA.
|
|
The food additive disodium EDTA (disodium ethylenediaminetetraacetate) may be safely used in designated foods for the purposes and in accordance with the following prescribed conditions:
(a) The additive contains a minimum of 99 percent disodium ethylenedia-minetetraacetate dihydrate (C10H14O8N2Na2-2H2O).
(b) It is used or intended for use as follows:
(1) Alone, in the following foods at not to exceed the levels prescribed, calculated as anhydrous calcium disodium EDTA:
| Food
| Limitation (parts per million)
| Use
|
|---|
| Aqueous multivitamin preparations | 150 | With iron salts as a stabilizer for vitamin B
12 in liquid multivitamin preparations.
| | Canned black-eyed peas | 145 | Promote color retention.
| | Canned kidney beans | 165 | Preservative.
| | Canned strawberry pie filling | 500 | Promote color retention.
| | Cooked sausage | 36 | As a cure accelerator with sodium ascorbate or ascorbic acid.
| | Dressings, nonstandardized | 75 | Preservative.
| | French dressing | 75 | Do.
| | Frozen white potatoes including cut potatoes | 100 | Promote color retention.
| | Gefilte fish balls or patties in packing medium |
1 50 | Inhibit discoloration.
| | Legumes (all cooked canned, other than black-eyed peas) | 165 | Promote color retention.
| | Mayonnaise | 75 | Preservative.
| | Ready-to-eat cereal products containing dried bananas |
2 315 | Promote color retention.
| | Salad dressing | 75 | Preservative.
| | Sandwich spread | 100 | Do.
| | Sauces | 75 | Do.
|
(2) With calcium disodium EDTA (calcium disodium ethylenediaminetetraacetate; calcium disodium (ethylenedinitrilo) tetraacetate), in the following foods at not to exceed, in combination, the levels prescribed, calculated as anhydrous C10H12O8N2CaNa2:
| Food
| Limitation (parts per million)
| Use
|
|---|
| Dressings, nonstandardized | 75 | Preservative.
| | French dressing | 75 | Do.
| | Mayonnaise | 75 | Do.
| | Salad dressing | 75 | Do.
| | Sandwich spread | 100 | Do.
| | Sauces | 75 | Do. |
(3) Alone, as a sequestrant in the nonnutritive sweeteners that are listed in § 180.37 of this chapter and that, in addition, are designed for aqueous solution: Provided, That the amount of the additive, calculated as anhydrous calcium disodium EDTA, does not exceed 0.1 percent by weight of the dry nonnutritive sweetener.
(c) To assure the safe use of the additive:
(1) The label and labeling of the additive container shall bear, in addition to the other information required by the act, the name of the additive.
(2) The label or labeling of the additive container shall bear adequate use directions to provide a final food product that complies with the limitations provided in paragraph (b) of this section.
(d) In the standardized foods listed in paragraphs (b)(1) and (2) of this section the additives are used only in compliance with the applicable standards of identity for such foods.
[42 FR 14491, Mar. 15, 1977, as amended at 65 FR 48379, Aug. 8, 2000]
|
|
|
(a) Ethoxyquin (1,2-dihydro-6-ethoxy-2,2,4-trimethylquinoline) may be safely used as an antioxidant for preservation of color in the production of chili powder, paprika, and ground chili at levels not in excess of 100 parts per million.
(b) In order to provide for the safe use of the additive in feed prepared in accordance with §§ 573.380 and 573.400 of this chapter, tolerances are established for residues of ethoxyquin in or on edible products of animals as follows:
5 parts per million in or on the uncooked fat of meat from animals except poultry.
3 parts per million in or on the uncooked liver and fat of poultry.
0.5 part per million in or on the uncooked muscle meat of animals.
0.5 part per million in poultry eggs.
Zero in milk.
|
|
|
Sec. 172.145 Heptylparaben.
|
|
(a) The food additive heptylparaben is the chemical n -heptyl p -hydroxybenzoate.
(b) It may be safely used to inhibit microbiological spoilage in accordance with the following prescribed conditions:
(1) In fermented malt beverages in amounts not to exceed 12 parts per million.
(2) In noncarbonated soft drinks and fruit-based beverages in amounts not to exceed 20 parts per million, when standards of identity established under section 401 of the Act (21 U.S.C. 341) do not preclude such use.
|
|
|
Sec. 172.150 4-Hydroxymethyl-2,6-di-tert-butylphenol.
|
|
The food additive 4-hydroxymethyl-2,6-di-tert -butylphenol may be safely used in food in accordance with the following prescribed conditions:
(a) The additive has a solidification point of 140 deg.C-141 deg.C.
(b) The additive is used as an antioxidant alone or in combination with other permitted antioxidants.
(c) The total amount of all antioxidants added to such food shall not exceed 0.02 percent of the oil or fat content of the food, including the essential (volatile) oil content of the food.
|
|
|
Sec. 172.155 Natamycin (pimaricin).
|
|
(a) Natamycin (CAS Reg. No. 7681-93-8), also known as pimaricin, is a polyene macrolide antimycotic substance possessing an empirical formula of C33H47NO13 and a molecular weight of 665.7.
(b) The additive shall conform to the following specifications:
Purity: 97 percent +/-2 percent on an anhydrous basis.
Arsenic: Not more than 1 part per million.
Heavy metals (as Pb): Not more than 20 parts per million.
(c) The additive may be applied on cheese, as an antimycotic, in amounts not to exceed 20 milligrams per kilogram (20 parts per million) in the finished product as determined by International Dairy Federation (IDF) Standard 140A:1992, "Cheese and Cheese Rind-Determination of Natamycin Content-Method by Molecular Absorption Spectrometry and by High-Performance Liquid Chromatography," which is incorporated by reference. The Director of the Office of the Federal Register approves this incorporation by reference in accordance with 5 U.S.C. 552(a) and 1 CFR part 51. Copies are available from the Office of Food Additive Safety (HFS-200), Center for Food Safety and Applied Nutrition, Food and Drug Administration, 5001 Campus Dr., College Park, MD 20740, 240-402-1200, or may be examined at the Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852, 240-402-7500, between 9 a.m. and 4 p.m., Monday through Friday, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030, or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html.
[47 FR 26823, June 22, 1982, as amended at 50 FR 49536, Dec. 3, 1985; 63 FR 66015, Dec. 1, 1998; 66 FR 13847, Mar. 8, 2001; 81 FR 5591, Feb. 3, 2016; 88 FR 17719, Mar. 24, 2023]
|
|
|
Sec. 172.160 Potassium nitrate.
|
|
The food additive potassium nitrate may be safely used as a curing agent in the processing of cod roe, in an amount not to exceed 200 parts per million of the finished roe.
|
|
|
Sec. 172.165 Quaternary ammonium chloride combination.
|
|
The food additive, quaternary ammonium chloride combination, may be safely used in food in accordance with the following conditions:
(a) The additive contains the following compounds: n- dodecyl dimethyl benzyl ammonium chloride (CAS Reg. No. 139-07-1); n- dodecyl dimethyl ethylbenzyl ammonium chloride (CAS Reg. No. 27479-28-3); n- hexadecyl dimethyl benzyl ammonium chloride (CAS Reg. No. 122-18-9); n- octadecyl dimethyl benzyl ammonium chloride (CAS Reg. No. 122-19-0); n- tetradecyl dimethyl benzyl ammonium chloride (CAS Reg. No. 139-08-2); n- tetradecyl dimethyl ethylbenzyl ammonium chloride (CAS Reg. No. 27479-29-4).
(b) The additive meets the following specifications: pH (5 percent active solution) 7.0-8.0; total amines, maximum 1 percent as combined free amines and amine hydrochlorides.
(c) The additive is used as an antimicrobial agent, as defined in § 170.3(o)(2) of this chapter, in raw sugar cane juice. It is added prior to clarification when further processing of the sugar cane juice must be delayed.
(d) The additive is applied to the sugar juice in the following quantities, based on the weight of the raw cane:
| Component
| Parts per million
|
|---|
| n-Dodecyl dimethyl benzyl ammonium chloride | 0.25-1.0
| | n-Dodecyl dimethyl ethylbenzyl ammonium chloride | 3.4-13.5
| | n-Hexadecyl dimethyl benzyl ammonium chloride | 1.5-6.0
| | n-Octadecyl dimethyl benzyl ammonium chloride | 0.25-1.0
| | n-Tetradecyl dimethyl benzyl ammonium chloride | 3.0-12.0
| | n-Tetradecyl dimethyl ethylbenzyl ammonium chloride | 1.6-6.5 |
[50 FR 3890, Jan. 29, 1985]
|
|
|
Sec. 172.167 Silver nitrate and hydrogen peroxide solution.
|
|
An aqueous solution containing a mixture of silver nitrate and hydrogen peroxide may be safely used in accordance with the following prescribed conditions:
(a) The additive is used as an antimicrobial agent in bottled water.
(b) Hydrogen peroxide meets the specifications of the Food Chemicals Codex, 7th ed. (2010), pp. 496-497, which is incorporated by reference. The Director of the Office of the Federal Register approves this incorporation by reference in accordance with 5 U.S.C. 552(a) and 1 CFR part 51. You may obtain copies from the United States Pharmacopeial Convention, 12601 Twinbrook Pkwy., Rockville, MD 20852 (Internet address http://www.usp.org ). Copies may be examined at the Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852, 240-402-7500, between 9 a.m. and 4 p.m., Monday through Friday, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030 or go to: http://www.archives.gov/federal-register/cfr/ibr-locations.html.
(c) The amount of silver added will not exceed 17 micrograms per kilogram in the treated bottled water, and the amount of hydrogen peroxide will not exceed 23 milligrams per kilogram in the treated bottled water. Analyses for silver and hydrogen peroxide shall be conducted on samples of treated bottled water at the site of bottling, using samples of the water intended for treatment for the blank determination.
(d)(1) The amount of silver in the treated bottled water is determined using the method for silver designated in 21 CFR 165.110(b)(4)(iii)(G)(2 )(i ).
(2) The amount of hydrogen peroxide in the treated bottled water is determined using a Hydrogen Peroxide Test Kit from the HACH Co., or equivalent. The manual from the Hydrogen Peroxide Test Kit, Model HYP-1, Catalog Number 22917-00, 1991, is incorporated by reference. The Director of the Federal Register approves this incorporation by reference in accordance with 5 U.S.C. 552(a) and 1 CFR part 51. You may obtain copies of the test kit manual from the HACH Co., P.O. Box 389, Loveland CO, 80359 (1-800-227-4224), Model HYP-1, Catalog Number 22917-00. Copies may be examined at the Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852, 240-402-7500, between 9 a.m. and 4 p.m., Monday through Friday, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030 or go to: http://www.archives.gov/federal_register/code_of_federal_regulations/ibr_locations.html.
(e) Substances generally recognized as safe in or on food may be used to stabilize the additive to ensure that the additive will perform its intended technical effect.
(f) The additive may not be added to bottled water that has been filtered or is intended to be filtered through a silver-containing water filter.
(g) Bottled water must meet the quality standards for bottled water in § 165.110(b)(2) through (b)(5) of this chapter, including the limits specified for total silver and nitrate, unless the water bears a label statement of substandard quality, as provided for under § 165.110(c) of this chapter.
[74 FR 11478, Mar. 18, 2009, as amended at 78 FR 71461, Nov. 29, 2013; 81 FR 5591, Feb. 3, 2016; 88 FR 17719, Mar. 24, 2023]
|
|
|
Sec. 172.170 Sodium nitrate.
|
|
The food additive sodium nitrate may be safely used in or on specified foods in accordance with the following prescribed conditions:
(a) It is used or intended for use as follows:
(1) As a preservative and color fixative, with or without sodium nitrite, in smoked, cured sablefish, smoked, cured salmon, and smoked, cured shad, so that the level of sodium nitrate does not exceed 500 parts per million and the level of sodium nitrite does not exceed 200 parts per million in the finished product.
(2) As a preservative and color fixative, with or without sodium nitrite, in meat-curing preparations for the home curing of meat and meat products (including poultry and wild game), with directions for use which limit the amount of sodium nitrate to not more than 500 parts per million in the finished meat product and the amount of sodium nitrite to not more than 200 parts per million in the finished meat product.
(b) To assure safe use of the additive, in addition to the other information required by the Act:
(1) The label of the additive or of a mixture containing the additive shall bear:
(i) The name of the additive.
(ii) A statement of the concentration of the additive in any mixture.
(2) If in a retail package intended for household use, the label and labeling of the additive, or of a mixture containing the additive, shall bear adequate directions for use to provide a final food product that complies with the limitations prescribed in paragraph (a) of this section.
(3) If in a retail package intended for household use, the label of the additive or of a mixture containing the additive, shall bear the statement "Keep out of the reach of children".
|
|
|
Sec. 172.175 Sodium nitrite.
|
|
The food additive sodium nitrite may be safely used in or on specified foods in accordance with the following prescribed conditions:
(a) It is used or intended for use as follows:
(1) As a color fixative in smoked cured tunafish products so that the level of sodium nitrite does not exceed 10 parts per million (0.001 percent) in the finished product.
(2) As a preservative and color fixative, with or without sodium nitrate, in smoked, cured sablefish, smoked, cured salmon, and smoked, cured shad so that the level of sodium nitrite does not exceed 200 parts per million and the level of sodium nitrate does not exceed 500 parts per million in the finished product.
(3) As a preservative and color fixative, with sodium nitrate, in meat-curing preparations for the home curing of meat and meat products (including poultry and wild game), with directions for use which limit the amount of sodium nitrite to not more than 200 parts per million in the finished meat product, and the amount of sodium nitrate to not more than 500 parts per million in the finished meat product.
(b) To assure safe use of the additive, in addition to the other information required by the Act:
(1) The label of the additive or of a mixture containing the additive shall bear:
(i) The name of the additive.
(ii) A statement of the concentration of the additive in any mixture.
(2) If in a retail package intended for household use, the label and labeling of the additive, or of a mixture containing the additive, shall bear adequate directions for use to provide a final food product which complies with the limitations prescribed in paragraph (a) of this section.
(3) If in a retail package intended for household use, the label of the additive, or of a mixture containing the additive, shall bear the statement "Keep out of the reach of children".
|
|
|
Sec. 172.177 Sodium nitrite used in processing smoked chub.
|
|
The food additive sodium nitrite may be safely used in combination with salt (NaCl) to aid in inhibiting the outgrowth and toxin formation from Clostridium botulinum type E in the commercial processing of smoked chub in accordance with the following prescribed conditions:
(a) All fish in smoking establishments shall be clean and wholesome and shall be expeditiously processed, packed, and stored under adequate sanitary conditions in accordance with good manufacturing practice.
(b) The brining procedure is controlled in such a manner that the water phase portion of the edible portion of the finished smoked product has a salt (NaCl) content of not less than 3.5 percent, as measured in the loin muscle, and the sodium nitrite content of the edible portion of the finished smoked product is not less than 100 parts per million and not greater than 200 parts per million, as measured in the loin muscle.
(c) Smoked chub shall be heated by a controlled heat process which provides a monitoring system positioned in as many strategic locations in the smokehouse as necessary to assure a continuous temperature throughout each fish of at least 160 deg.F for a minimum of 30 minutes.
(d) The finished product shall be cooled to a temperature of 50 deg.F or below within 3 hours after smoking and further cooled to a temperature of 38 deg.F or below within 12 hours after smoking. A temperature of 38 deg.F or below shall be maintained during all subsequent storage and distribution. All shipping containers, retail packages, and shipping records shall indicate with appropriate notice the perishable nature of the product and specify that the product shall be held under refrigeration (38 deg.F or below) until consumed.
(e) To assure safe use of the additive:
(1) The label and labeling of the additive container shall bear, in addition to the other information required by the Act, the name of the additive.
(2) The label or labeling of the additive container shall bear adequate directions to assure use in compliance with the provisions of this section.
|
|
|
Sec. 172.180 Stannous chloride.
|
|
The food additive stannous chloride may be safely used for color retention in asparagus packed in glass, with lids lined with an inert material, in an amount not to exceed 20 parts per million calculated as tin (Sn).
|
|
|
The food additive TBHQ, which is the chemical 2-(1,1-dimethylethyl)-1,4-benzenediol (Chemical Abstracts Service Registry Number 1948-33-0), also known as tertiary butylhydroquinone, may be safely used in food in accordance with the following prescribed conditions:
(a) The food additive has a melting point of not less than 126.5 deg.C.
(b) The percentage of TBHQ in the food additive is not less than 99.0 percent when tested by the assay described in the Food Chemicals Codex, 9th ed. (2014), pp. 1192-1194, which is incorporated by reference, or an equivalent method. The Director of the Office of the Federal Register approves this incorporation by reference in accordance with 5 U.S.C. 552(a) and 1 CFR part 51. You may obtain copies from the United States Pharmacopeial Convention, 12601 Twinbrook Pkwy., Rockville, MD 20852 (Internet address: http://www.usp.org ). Copies may be examined at the Dockets Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852, 240-402-7500, between 9 a.m. and 4 p.m., Monday through Friday, or at the National Archives and Records Administration (NARA). For information on the availability of this material at NARA, call 202-741-6030 or go to: http://www.archives.gov/federal-register/cfr/ibr-locations.html .
(c) It is used as an antioxidant alone or in combination with BHA and/or BHT.
(d) The total antioxidant content of a food containing the additive will not exceed 0.02 percent of the oil or fat content of the food, including the essential (volatile) oil content of the food.
[42 FR 14491, Mar. 15, 1977, as amended at 80 FR 34276, June 16, 2015; 88 FR 17719, Mar. 24, 2023]
|
|
|
The food additive THBP (2,4,5-trihydroxybutyrophenone) may be safely used in food in accordance with the following prescribed conditions:
(a) The food additive has a melting point of 149 deg.C-153 deg.C.
(b) It is used as an antioxidant alone or in combination with other permitted antioxidants.
(c) The total antioxidant content of a food containing the additive will not exceed 0.02 percent of the oil or fat content of the food, including the essential (volatile) oil content of the food.
|
|
|
Authority: 21 U.S.C. 321, 341, 342, 348, 371, 379e.
Source: 42 FR 14491, Mar. 15, 1977, unless otherwise noted.
|
|